
In the same plane, each F-Br-F bond forms a 90-degree angle. Because the core central atom, bromine, has five Br-F bonds with the surrounding fluorine atoms. In polar protic solvents like aqueous KOH, as OH- is the nucleophile and as H+ is present in water. What are BrF5 electron and molecular geometry BrF5 has a square pyramidal molecular geometry and an octahedral electron geometry, according to the VSEPR theory. Is Koh Protic or aprotic?īecuase, polar aprotic solvents are used in SN2 reaction, because they do not effect the nucleophile in rate determine step. The electron-pair geometry provides a guide to the bond angles of. In each case, predict (a) the approximate bond angle(s), (b) the hybridization around the underlined. Make a sketch of BrF5, using the bond conventions shown in the Box in Section 10.4 in the textbook. Predicting Molecular Geometry and Hybridization. Ether, methylene chloride, and hexane are examples of aprotic solvents. Molecular geometry is the name of the geometry used to describe the shape of a molecule. Determine the molecular geometry of BrF5. Solvents that cannot act as hy- drogen-bond donors are called aprotic solvents. Water, alcohols, and carboxylic acids are examples of protic solvents. Are ethers Protic or aprotic?Ī protic solvent consists of molecules that can act as hydrogen-bond donors. What is the bond angle of SiH4?Īlthough the four bonds C-Cl are polar because of the difference in electronegativity of Chlorine(3.16) and Carbon(2.55), CCl4 is nonpolar because the bond polarity gets canceled with each other due to the symmetrical geometrical structure (tetrahedral) of the CCl4 molecule.ĭimethylformamide is a polar aprotic solvent because it is a polar molecule and has no OH or NH groups.

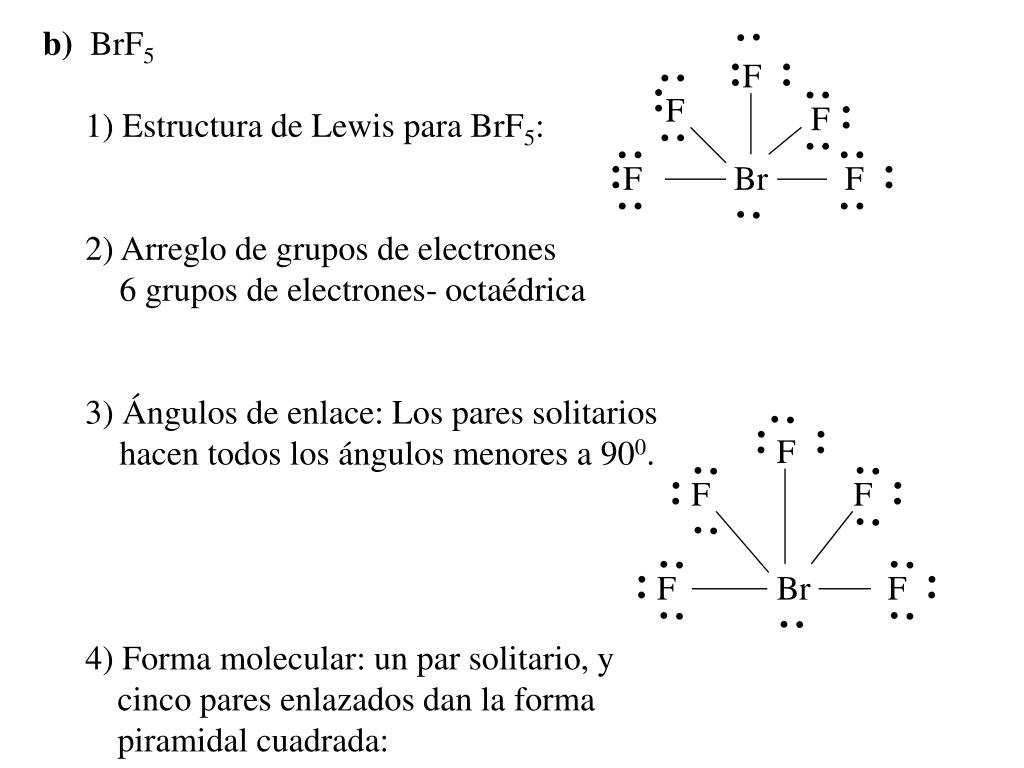
The sp3d2 hybridization of the BrF5 molecule is formed when one S orbital, three p orbitals, and two d orbital join together to form the BrF5 molecular orbital. The bromine and fluorine atoms have s,p and d orbitals. The bigger molecular weight of SiH4 than CH4 makes the SiH4 molecule larger than CH4. The BrF5 molecule ion hybridization is six. Quiz your students on Lewis Structure For BrF5, Molecular Geometry, Bond Angle, Hybridization, Polar or Nonpolar using our fun classroom quiz game Quizalize. This means that both CH4 and SiH4 have very low ΔEN which is sufficiently weak to have permanent dipole-dipole intermolecular attraction. Because of the lobe pair effect, the structure arranges itself in square planar geometry. The molecular geometry of SiH4 is tetrahedral with symmetric charge distribution around the central atom. It has a square planar geometry as the 7 outermost electrons of bromine form bonds with 5 F atoms leaving behind a lone pair of electrons.
